ACECLOFENAC VS DICLOFENAC
Trials of 2 to 6 months duration have shown Aceclofenac to be an effective agent in the management of pain and rheumatic disease. Data from in vitro studies indicate properties of particular interest with respect to cartilage matrix effects and selectivity for COX-2. Aceclofenac is well tolerated, with encouraging reports of improved general and GI tolerability relative to other NSAIDs from a meta-analysis of double-blind trials and from large non blind studies. Superior overall and/or GI tolerability of the drug relative to other NSAIDs has been indicated by a non randomized comparison with sustained release Diclofenac in 10,142 patients, a meta-analysis of 13 comparisons with Diclofenac, naproxen, piroxicam, indomethacin, tenoxicam or ketoprofen in 3574 patients, and preliminary details of a comparison with 10 other NSAIDs in 142,776 patients [9].
A pan-European study involving 23407 patients with pain due to various inflammatory or degenerative rheumatic diseases was undertaken in Austria, Belgium, Germany and Greece, to evaluate overall pain relief and satisfaction with Aceclofenac therapy. Aceclofenac was considered by patients to be a highly efficacious treatment with excellent and fast analgesic activity that was maintained throughout the study period. At the conclusion of the study, assessment of patient status, a parameter encompassing both efficacy against inflammatory pain and tolerability, by both patient and physician, was either much improved or improved in 84% of cases. These evaluations were similar irrespective of the country or whether the indication was acute (e.g. post-pain) or chronic pain (e.g. osteoarthritis) [12].
Patient satisfaction with, and compliance of, Aceclofenac therapy was similarly impressive; 90% of patients were satisfied and over 90% of patients were treatment compliant. In combination with the recently published SAMM study results, the findings of the European Observational Cohort study validate Aceclofenac, in everyday clinical practice, as an effective, well-tolerated and well-accepted therapy for both acute and chronic inflammatory and degenerative disease. The availability of a powerful anti-inflammatory agent with a low incidence of side-effects is of considerable value to both the patient and physician in the management of inflammatory pain. This objective has been fulfilled with Aceclofenac therapy [12].
The result of a multicentre randomised, double-blind, parallel group confirmed that Aceclofenac offers greater improvement in complete knee movement and reduced pain on movement. This study investigate the efficacy and safety of Aceclofenac (200 patients, 100 mg twice daily and placebo once daily) in comparison with Diclofenac (197 patients, 50mg three times daily) in patients with osteoarthritis of the knee. The treatment period of twelve weeks was preceded by a washout period of two weeks duration
In patients with initial flexion deformity, Aceclofenac was significantly more effective than Diclofenac in improving knee flexion after 2-4 weeks treatment. Patients’ subjective assessment of pain relief demonstrated significantly greater efficacy with Aceclofenac. At end point, 71% of patients in the Aceclofenac group reported improvement in pain intensity as compared to 59% treated with Diclofenac [13].
At end point, slightly better clinical assessment has been observed with Aceclofenac as compared with Diclofenac which includes joint tenderness, swelling, pain on movement, functional capacity, overall assessment. Aceclofenac and Diclofenac-treated groups both exhibited significant improvement in pain intensity [13].
Aceclofenac has better tolerability profile than with Diclofenac as fewer patients experienced gastrointestinal adverse events. In particular, the incidence of treatment related diarrhea is less with Aceclofenac (1%) than the Diclofenac (6.6%) [13].
THERAPEUTIC ADVANTAGEOUS ACHNIL VS OTHER ACECLOFENAC PREPARATIONS
- Patented in INDIA and EUROPE
- Once a day preparation of first time in the world
- Immediate action with in 4-5 min due to immediate Burst release
- Maintain effective Plasma concentration for 24 hrs above the therapeutic Window
- Superior Anti-inflammatory action due to multi factorial mechanism of action
- Greater Efficacy as works for 24 hrs due to round the clock the inhibition of nociceptive stimuli
- Very Less painful due to very less viscosity
- Stable as no precipitation is there on the storage of drug
- Safe and non toxic injectable formulation
- Improved patient compliance
- Clinically Aceclofenac is highly superior to Diclofenac
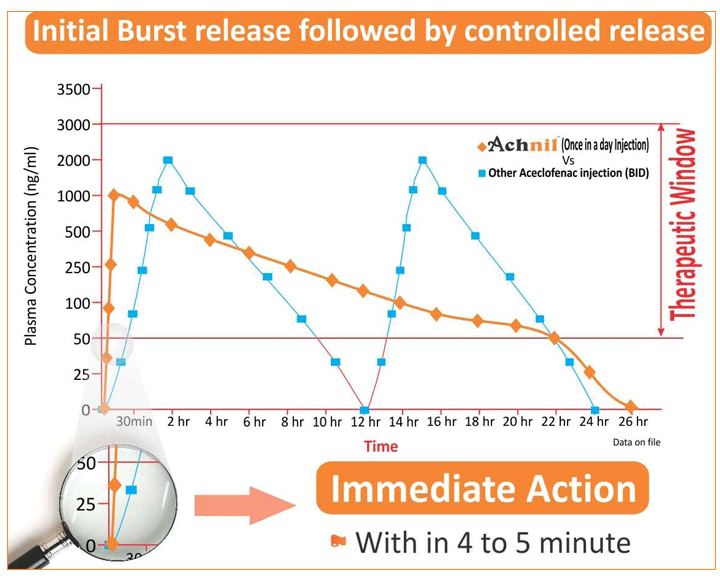
As shown in the graph initially ACHNIL immediately release approximately 20 % of the drug thus shows onset of action within 45 minutes. Then Followed the controlled release pattern up to 24 hrs which shows long action hence dose is OD Whereas other ACECLOFENAC preparations acts only for 10-12 hrs hence dose is BD
ACHNIL VS DICLOFENAC
OBJECTIVE
To evaluate the efficacy and tolerability of Aceclofenac injection as compared to Diclofenac injection in subjects suffering from acute lumbago.
NUMBER OF PATIENTS
A total of 300 patients were randomly attributed assigned to receive either Aceclofenac (150 patients) or Diclofenac (150 patients).
INTERVENTIONS
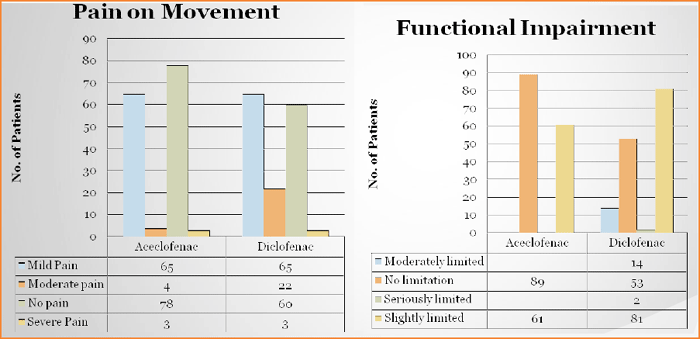
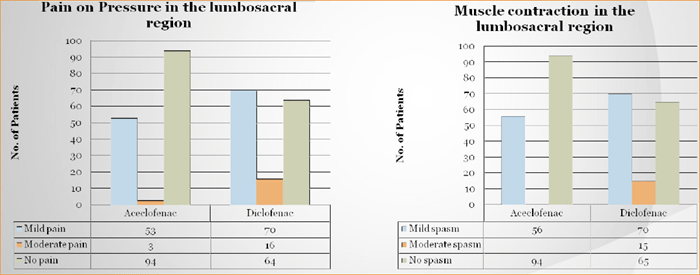
Study has been conducted on the parameters of Pain on movement, Pain on pressure, Functional impairment, Muscle contraction.
RESULTS
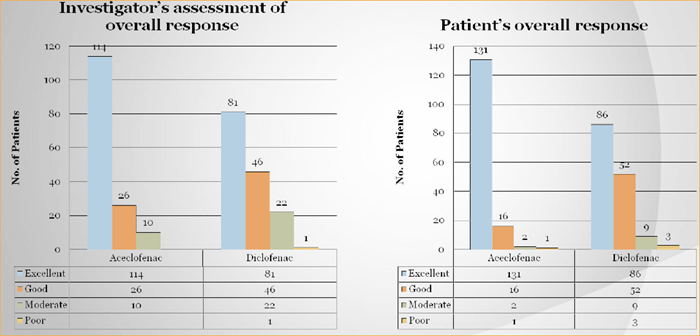
Both groups showed improvement on VAS for pain at rest at the end of 30 minutes, 1, 2, 4, 8, 24 and 48 hours after treatment (Table 1). Aceclofenac showed a quantitatively better improvement in the change in mean VAS score from baseline at all points of measurement. This improvement became statistically in favor of Aceclofenac group at all points of assessment (p<0.05).
| Table 1: Changes in mean VAS score from baseline | |||||||
|---|---|---|---|---|---|---|---|
| Group | 30 min | 1 hr | 2 hr | 4 hr | 8 hr | 24 hr | 48 hr |
| Aceclofenac (n=150) | |||||||
| Mean | 13.37 | 21.35 | 29.42 | 37.12 | 43.06 | 49.46 | 55.77 |
| SD | 6.91 | 7.89 | 9.01 | 1016 | 10.40 | 10.14 | 8.79 |
| Diclofenac (n=150) | |||||||
| Mean | 11.06 | 19.06 | 27.78 | 36.50 | 42.10 | 48.00 | 52.95 |
| SD | 4.57 | 6.21 | 7.20 | 7.37 | 8.70 | 9.92 | 11.33 |
| t-test p value | P<0.05 | P<0.05 | P<0.05 | P<0.05 | P<0.05 | P<0.05 | P<0.05 |
The modified Schober’s test showed progressive improvements at 8 hrs, 24 hrs and after 48 hrs of treatment in both groups, but the improvement was higher and significant in the Aceclofenac group than in the Diclofenac at 8 hrs, 24 hrs and after 48 hrs of treatment (Table 2).
| Table 2: Changes in mean Schober’s test value from baseline | |||
|---|---|---|---|
| 8 hr | 24 hr | 48 hr | |
| Aceclofenac (n=150) | |||
| Mean | -0.061 | -1.09 | -2.36 |
| SD | 0.34 | 0.50 | 5.70 |
| Diclofenac (n=150) | |||
| Mean | -0.47 | -0.87 | -1.36 |
| SD | 0.26 | 0.42 | 0.75 |
| t-test p value | PP<0.05P<0.05 | ||
CONCLUSION: Achnil (Aceclofenac injection) OD was superior to Diclofenac BID